Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 solid solutions
solid solutionsMorimoto, Kyoichi; Kato, Masato; Ogasawara, Masahiro*; Kashimura, Motoaki; Abe, Tomoyuki
Journal of Alloys and Compounds, 452(1), p.54 - 60, 2008/03
Times Cited Count:30 Percentile:77.75(Chemistry, Physical)Plutonium and uranium mixed oxide (MOX) fuel with high Pu content have been developed as a fuel of fast reactor (FR). As the storage time of Pu raw material between reprocessing and fabrication increases, americium content of the fabricated MOX fuel increases up to a few percent. In this work, the thermal conductivity of MOX fuel containing Am was investigated as a part of clarifying the effect of Am content on thermal physical properties. The pellets of (Am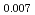 Pu
Pu U)O
U)O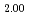 , (Am
, (Am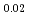 Pu
Pu U)O
U)O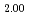 and (Am
and (Am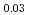 Pu
Pu U)O
U)O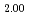 were prepared. The oxygen to metal ratio (O/M ratio) of sintered pellet was adjusted to 2.00. The thermal diffusivity measurement was carried out in the range of temperature from 900 K to 1700 K by the laser flash method, and thermal conductivity of these pellets was evaluated. The heat capacity for evaluating thermal conductivity was derived from heat capacity of UO
were prepared. The oxygen to metal ratio (O/M ratio) of sintered pellet was adjusted to 2.00. The thermal diffusivity measurement was carried out in the range of temperature from 900 K to 1700 K by the laser flash method, and thermal conductivity of these pellets was evaluated. The heat capacity for evaluating thermal conductivity was derived from heat capacity of UO , PuO
, PuO and AmO
and AmO by using the Kopp-Neumann rule.
by using the Kopp-Neumann rule.
Kato, Masato; Morimoto, Kyoichi; Sugata, Hiromasa*; Konashi, Kenji*; Kashimura, Motoaki; Abe, Tomoyuki
Journal of Alloys and Compounds, 452(1), p.48 - 53, 2008/03
Times Cited Count:30 Percentile:77.75(Chemistry, Physical)Plutonium and uranium mixed oxide has been developed as a fuel of a fast reactor. The maximum temperature of the fuel pellet is limited within a design criterion to prevent fuel melting. So, the melting points of the mixed oxide have been investigated since the development of fast reactor started. However the measured data are limited. In this work, the melting points of (U1-yPuy)O (y: 0, 0.12, 0.2, 0.3, 0.4) were measured by the thermal arrest method. The evaluated melting point of this study underestimates in case of MOX with high Pu contents of 30% and 40%. The solidus of UO
(y: 0, 0.12, 0.2, 0.3, 0.4) were measured by the thermal arrest method. The evaluated melting point of this study underestimates in case of MOX with high Pu contents of 30% and 40%. The solidus of UO , (Pu
, (Pu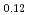 U
U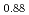 )O
)O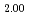 and (Pu
and (Pu U
U )O
)O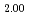 were determined to be 3128K, 3077K and 3052K, respectively. The solidus temperature of hypostoichiometric MOX slightly increased with decreasing O/M.
were determined to be 3128K, 3077K and 3052K, respectively. The solidus temperature of hypostoichiometric MOX slightly increased with decreasing O/M.
 Ln
Ln O
O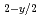 (Ln = lanthanide; M = Hf, Zr, Ce, U, Th); Structure, property, and applications
(Ln = lanthanide; M = Hf, Zr, Ce, U, Th); Structure, property, and applicationsNakamura, Akio; Masaki, Nobuyuki; Otobe, Haruyoshi; Hinatsu, Yukio*; Wang, J.*; Takeda, Masuo*
Pure and Applied Chemistry, 79(10), p.1691 - 1729, 2007/10
Times Cited Count:14 Percentile:47.07(Chemistry, Multidisciplinary)no abstracts in English